Physical Properties of Substances
|
Let us take this graduated cylinder filled with water. It has the same physical properties as the water below. Even though the amount of liquid in the graduated cylinder is less than the amount of water in the PYREX jug, they will always have the same physical properties.
The same principal applies with solids too. It doesn't matter how much of a certain substance there is. Their physical properties will always be the same. Also if there is a good conductor of heat, it will always be a good conductor of electricity. For example, let us take copper. Copper is a good conductor of electricity. Because it is a good conductor of electricity, it will be a good conductor of heat as well. If I take 20 meters of copper, as shown below, then it will have the same physical properties as an amount of copper that is more or less than 20 meters. It will work with any amount of a certain solid or liquid or gas of the same type. They will have the same physical properties. DENSITY As defined above density is mass/volume. Density is normally defined as a comparison between liquids. If one colored liquid is on the bottom of a graduated cylinder and another colored liquid is on the top of the graduated cylinder, then the liquid on the bottom is more dense than the liquid on the bottom. An image of this happening is shown below. The yellow liquid is the least dense because it is on the top of the other liquids. The most dense liquid is the maroon colored liquid, which is on the bottom.
SOLUBILITY
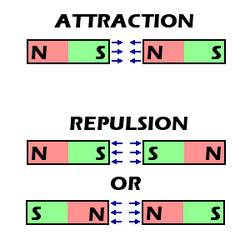
Solubility is the ability of a specific solid to dissolve. Salt can dissolve into water very easily because it is very soluble. Magnetism If I had two magnets and I point the North part of one magnet to the south part of the other magnet, the magnets will attract each other. However if i point the north end of one magnet to the north end of another magnet, then the magnets will repel. The example below shows magnets repelling and attracting. |
SC.8.P.8.4: Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured: for example, density; thermal or electrical conductivity; solubility; magnetic properties; melting and boiling points; and know that these properties are independent of the amount of the sample.
Summary of Standard: Compare substances based on their physical properties and know that these properties are independent of the amount of a certain amount of that substance. Vocabulary Substance - Any solid, liquid, or gas Density - Normally defined as mass/volume. The symbol used for it is ρ (lower case Greek letter rho). The unit for it is kg/m^3. Thermal conductivity - A physical property of any material. It is the property for that material to conduct heat. Electrical conductivity - A physical property of any material that defines it's ability to conduct electricity. Solubility - A property of any solid, or liquid. It is the ability of that certain solid to dissolve. Magnetism - The force between two magnets. They either attract or repel each other. Melting point - The set temperature of a solid at which the solid liquefies. Boiling point - The set temperature at which a liquid turns into a gas. Liquids of the same substance have the same physical properties no matter what their mass is. |
Sources for pictures (in order that they appear) :
http://www.chemistryland.com/CHM107Lab/Lab7/Slime/Lab7Slime.htm
http://mad-science.wonderhowto.com/how-to/sweet-smell-success-diy-smoke-mix-with-sugar-and-potassium-nitrate-0134482/
http://www.alchemyandice.com/pure-copper-craft-wire-26g-0.40mm-20-metres.html
http://en.wikipedia.org/wiki/Density
https://www.kjmagnetics.com/blog.asp?p=magnet-basics
http://www.chemistryland.com/CHM107Lab/Lab7/Slime/Lab7Slime.htm
http://mad-science.wonderhowto.com/how-to/sweet-smell-success-diy-smoke-mix-with-sugar-and-potassium-nitrate-0134482/
http://www.alchemyandice.com/pure-copper-craft-wire-26g-0.40mm-20-metres.html
http://en.wikipedia.org/wiki/Density
https://www.kjmagnetics.com/blog.asp?p=magnet-basics